SOPHIA ANTIPOLIS, France – November 15, 2023 │ KnowMade’s Therapeutic mRNA Patent Monitor was presented at the 11th mRNA health conference in Berlin (October 31 to November 2, 2023) through the poster N°49 (High resolution PDF download here).
Abstract
Therapeutic mRNA, which became widely known as vaccine against COVID-19, are expected to be applicated to various areas such as cancer and regenerative medicine. In this fast-evolving context, monitoring the patenting activity related to therapeutic mRNA is crucial to be aware of weak signals, that might be the future of this technology, and understand the intellectual property position and strategy of the different players. Indeed, patent analysis is a relevant tool in market intelligence to anticipate new technological trends and future market competitions.
KnowMade has developed a strong expertise in therapeutic mRNA that allows detailed pictures of this disruptive technology, thanks to landscape reports on mRNA vaccine, mRNA cancer therapies and, more recently, self-amplifying RNA (SA RNA) vaccine. From these starting points, the monitoring activity follows, for each quarter, IP dynamics comprising new inventions, new granted IP rights and new litigations (US and Europe). The fine-tuned segmentation of KnowMade’s monitoring activity allows a deep understanding of technological sectors of current interest in therapeutic mRNA innovation. Indeed, this survey covers mRNA design (self-amplifying and circular RNA), carriers (lipid-based, polymeric, cell derived and inorganic), manufacturing & storing process, but also application and therapeutic area. The poster introduces some of the main dynamics since Q4 2022, such as tendencies in delivery strategy and arising of SA RNA approach.
Therapeutic mRNA’s IP monitoring activity has already shown in the past three quarters, that delivery is the most active technical segment and is highly dominated by LNP delivery systems, with more than 118 patent applications newly published in Q2 2023, 57 in Q1 2023 and 41 in Q4 2022 related to such nanoparticles (for more information read our press release Q2 2023, Q1 2023, and Q4 2022). LNP is the delivery system approved for COVID-19 vaccines and also subject of major litigations between main players such as Alnylam, Moderna and Pfizer, illustrating the importance of this technological aspect in therapeutic mRNA industry (for more information read our press release “Alnylam kicks off its third round in mRNA delivery battle”, June 06, 2023). Nevertheless, on the past quarters, its proportion in new patent publication decreased (from 70% in Q4 2022 to 56% in Q2 2023) while other systems, such as extracellular vesicles and polymeric nanoparticle, show positive dynamics in their development. Alternatives to LNPs is one of the solutions developed to overcome their main drawback, i.e., LNP accumulation in the liver limiting the access to other organs wherein the therapy is needed.
Beyond LNP IP activity, therapeutic mRNA’s IP monitoring activity answer the three-way questions “What? How? Who?”.
- WHAT – Q2-2023 activity by type of application, showing a growing interest in therapeutic drugs strategy.
- HOW – Self Amplifying mRNA IP dynamics overs past three quarters
- WHO – Current main players & Newcomers in the field
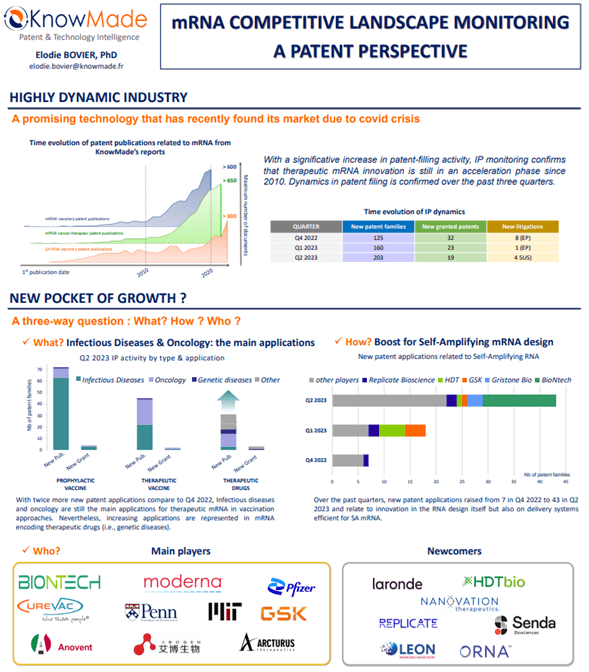
Poster #49 “mRNA Competitive landscape monitoring: A patent perspective” presented at the 11th mRNA health conference in Berlin. A high resolution PDF file can be downloaded for free here.
If you need more information, reach us at contact@knowmade.fr or with our contact forms.
Press contact
contact@knowmade.fr
Le Drakkar, 2405 route des Dolines, 06560 Sophia Antipolis, France
www.knowmade.com
About the author
Elodie Bovier, PhD., works at KnowMade as a Patent Analyst in the field of Biotechnology and Life Sciences. She holds a PhD in genetic and molecular biology from the Paris Sud University. She also holds the Industrial Property International Studies Diploma (in Patent and Trademark & Design Law) from the CEIPI (Strasbourg, France).
About KnowMade
KnowMade is a Technology Intelligence and IP Strategy consulting company specialized in analyzing patents and scientific publications. The company helps innovative companies, investors, and R&D organizations to understand their competitive landscape, follow technological evolutions, reduce uncertainties, and identify opportunities and risks in terms of technology and intellectual property.
KnowMade’s analysts combine their strong technology expertise and in-depth knowledge of patents with powerful analytics tools and methodologies to turn patent information and scientific literature into actionable insights, providing high added value reports for decision makers working in R&D, innovation strategy, intellectual property, and marketing. Our experts provide prior art search, patent landscape analysis, freedom-to-operate analysis, IP due diligence, and monitoring services.
KnowMade has a solid expertise in Compound Semiconductors, Power Electronics, Batteries, RF Technologies & Wireless Communications, Solid-State Lighting & Display, Photonics, Memories, MEMS & Sensors, Semiconductor Packaging, Medical Devices, Medical Imaging, Microfluidics, Biotechnology, Pharmaceutics, and Agri-Food.
