SOPHIA ANTIPOLIS, France – March 2nd, 2026 │ Wugen has received Breakthrough Therapy Designation from the U.S. FDA for its investigational CAR-T cell therapy, Sofi-cel, for patients with relapsed or refractory (R/R) T cell acute lymphoblastic leukemia or T cell lymphoblastic lymphoma (T-ALL/LBL). In these cancers, relapses remain a major challenge. These aggressive T-cell malignancies can progress rapidly, and outcomes after relapse are often poor. Sofi-cel or WU-CART-007 has already received multiple regulatory designations from the FDA such as regenerative medicine advanced therapy, fast track, orphan drug and rare pediatric disease designations, as well as priority medicines designation in the European Union. These recognitions reflect sustained regulatory confidence in the therapy’s potential.
Wugen at a Glance: An Off-the-Shelf CAR-T Strategy Supported by FDA Designations
Wugen was founded in 2018 in St. Louis, USA. It is a clinical-stage biotechnology company focused on developing allogeneic CAR-T cell therapies for cancer treatment. Wugen’s proprietary gene-editing platform is designed to overcome key limitations of first-generation cell therapies, enabling scalable off-the-shelf treatments.
Sofi-cel, or WU-CART-007, is an allogeneic anti-CD7 CAR-T cell therapy with CRISPR/Cas9 deletion of CD7 and T-cell receptor alpha constant (TRAC), to prevent fratricide and enable the use of healthy donor allogeneic T-cells with reduced risk of graft vs. host disease, respectively. CD7 is a transmembrane glycoprotein expressed in more than 95% of T-ALL and T-LBL cases, as well as in a subset of peripheral T-cell lymphomas. However, in healthy individuals, CD7 is expressed in around 90% of normal T cells and natural killer cells.
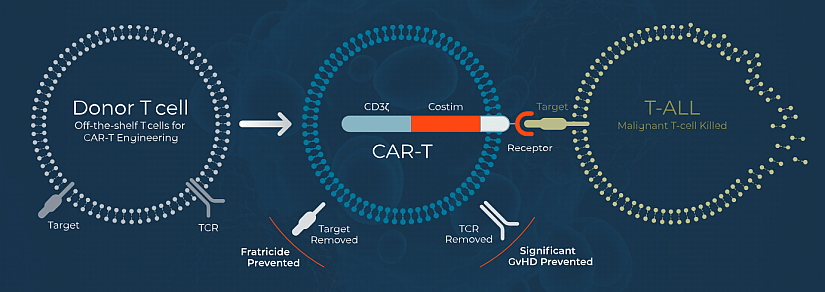
Figure: Wugen’s CAR-T platform (from Wugen website).
WU-CART-007 has already received Regenerative Medicine Advanced Therapy (RMAT), Fast Track, Orphan Drug, and Rare Pediatric Disease designations from the U.S. FDA and Priority Medicines (PRIME) Scheme designation in the European Union for the treatment of R/R T-ALL and T-LBL. In 2026, the allogeneic anti-CD7 CAR-T cell therapy obtained Breakthrough Therapy, a designation requested by the drug company. Breakthrough Therapy designation is “a process designed to expedite the development and review of drugs that are intended to treat a serious condition and preliminary clinical evidence indicates that the drug may demonstrate substantial improvement over available therapy on a clinically significant endpoint(s)”. The request should be received by FDA no later than the end-of-phase-2 meetings. The objective of this designation is to develop evidence needed to support approval as efficiently as possible. Breakthrough Therapy designation is different from Fast Track which “is a process designed to facilitate the development and expedite the review of drugs to treat serious conditions and fill an unmet medical need”. The goal is to get new drugs to the patient earlier. Fast Track designation must be requested by the company, at the same time, or any time after, the application submission for the investigation study of the drug.
Details of the WU-CART-007 Clinical Trials
The company has set up 4 clinical trials for WU-CART-007 against cancer. From two of them, there is no preliminary data disclosed. The NCT05509855 trial is an observational study, enrolling patients only by invitation, and the NCT05377827 is a phase 1 dose-escalation and dose-expansion study to evaluate the safety and tolerability of anti-CD7 allogeneic CAR T-Cells in patients with CD7+ hematologic malignancies.
The purpose of the NCT04984356 (January 2022) study is to evaluate the safety, recommended dose, and preliminary anti-tumor activity of WU-CART-007 in patients with relapsed or refractory T-cell acute lymphoblastic leukemia or lymphoblastic lymphoma. This is a first-in-human, multicenter, Phase 1, single-arm study in patients with R/R T-ALL/T-LBL who have exhausted other treatment options. The study evaluates dose escalation and dose expansion. There is a single intravenous infusion of WU-CART-007 cells on day 1 after lymphodepletion. Of 28 patients enrolled, 13 received the recommended phase 2 dose (RP2D) of 900 × 106 cells with enhanced lymphodepletion.
Preliminary data revealed by Wugen (article / presentation):
- Safety: The most common treatment-related adverse event was cytokine release syndrome. There are two grade 1 immune effector cell–associated neurotoxicity syndrome events and one grade 2 acute graft-versus-host disease event occurred. One grade 2 immune effector cell–associated hemophagocytic lymphohistiocytosis–like syndrome was observed.
- Efficacy: The overall response rate was 90.9%, and the composite complete remission rate was 72.7% for the 11 patients evaluable for response at the RP2D who received enhanced lymphodepleting chemotherapy.
According to these preliminary results, WU-CART-007 appears to have a manageable safety profile and a promising efficacy in heavily pretreated patients with relapsed/refractory T-ALL/LBL.
In January 2025, Wugen launched a phase 2 study, NCT06514794. This global T-RRex study enrolls pediatric and adult patients (1 year and older). It evaluates the efficacy of WU-CART-007 for patients with R/R T-ALL/LBL and WU-CART-007 as a therapy to induce complete Minimum Residual Disease (MRD) negative response. This is a Phase 2, single-arm, multi-center, open label study in 125 patients with R/R T-ALL/LBL and T-ALL/LBL in remission but remaining MRD positive. There is a single intravenous infusion of WU-CART-007 on day 1 after lymphodepletion.
Identified patent families related to WU-CART-007 clinical trial
Wugen owns 5 patent families (14 documents), filed between 2019 and 2022. The company has a worldwide IP strategy, with patent applications in Europe, the USA and Asia. Two-thirds of its patents are currently alive (pending applications and granted patents), reflecting that its R&D efforts are still on going to improve the current technologies involving cancer therapies. The strength of its patent portfolio may increase slightly in the coming years because 100% of its live patents are pending applications.
There is one patent family, filed in 2021, which relates CAR-T cells targeting CD7 (WO2022/098756). However, it is a lapsed patent family. A lapsed patent or patent application is not enforceable anymore because the applicant withdrew his patent application, failed to respond to an office action during the examination, or did not pay the maintenance fees. Typical office status for Lapsed could be “abandoned”, “lapsed”, “withdrawn”, etc.
The patent family described a method of treatment with a single dose, intravenously, of CD7 CAR-T cells. UCART7 is an allogeneic, fratricide-resistant, genetically modified T-cell transduced with a CAR having a CD28 costimulatory domain, and/or a 4-1BB costimulatory domain, and/or a CD3-zeta effector domain targeting CD7.
In example, a first-in-human Phase 1 study is described. It is a multicenter, dose-escalating, single- agent study conducted in patients with relapsed/refractory CD7-associated hematological cancers for which the investigator determines there is no other therapies available. All patients have failed multiples lines of conventional therapy applicable to their disease.
In part A of the study, the dose escalation proceeds independently in three groups: (1) Adult T-ALL/LBL, (2) adult T-NHL, (3) Pediatric T-ALL. Enrollment in the pediatric T-ALL group will begin after acceptable safety of the first dose level has been demonstrated in the adult T-ALL group. Upon attaining a Recommended Phase 2 Dose (RP2D), Part B of the study (cohort expansion) will commence. The RP2D may differ for T-ALL/LBL, adult T- NHL and pediatric T-ALL cohorts. In Part B of the study, each disease cohort (T-ALL/LBL, adult T-NHL, and pediatric T-ALL) will be expanded to explore a total of 15 patients per cohort. Part B of the study will seek to confirm the applicable dose for each patient subgroup and early signals of efficacy.
The relevance of animal models to predict an appropriate starting dose of UCART7 is limited so the dose levels selected have been based in part on existing clinical data derived from commercially available precedent autologous CAR-T therapies (which are not necessarily equivalent):
- Axicabtagene ciloleucel: The target dose is 2×106 CAR-T cells per kg body weight.
- Tisagenlecleucel: A single dose of 0.2 to 5.0×106 CAR- T cells per kg of body weight for patients 50 kg or less, or 0.1 to 2.5×108 CAR- T cells for patients more than 50 kg.
| Dose level | Cells/kg (patients<50 kg) | Fixed dose (patients>50 kg) | Fold increase form previous dose |
| DL1 | 2.0 x 106 | 100 x 106 | NA |
| DL2 | 6.0 x 106 | 300 x 106 | 3 |
| DL3 | 12.0 x 106 | 600 x 106 | 2 |
| DL4 | 18.0 x 106 | 900 x 106 | 1.5 |
Table: The proposed dose levels are shown in the table above.
All patients entering this study receive a single dose of UCART7 by intravenous (IV) injection at a dose level based on data evaluation from the prior cohort(s). Patients are admitted to the hospital and receive treatment, lymphodepleting chemotherapy followed by a UCART7 infusion. For UCART7 administration, patients receive UCART7 over 30 minutes at the designated IV dose 72 hours following Day 3 of the lymphodepletion protocol (i.e., on Day 0), providing they meet the following criteria: (a) no evidence of infection; (b) blood oxygenation within range of inclusion criteria; (c) no major complications of preconditioning lymphodepletion regimen. Patients will remain as inpatients for 7 days following the administration of UCART7. No preliminary data is available.
Sofi-cel’s Acceleration: Regulatory Endorsement, Ongoing Trials, and an Emerging IP Strategy
Wugen is a U.S.-based clinical-stage biotechnology company advancing off-the-shelf allogeneic CAR-T therapies for cancer, led by Sofi-cel (WU-CART-007), an anti-CD7 gene-edited CAR-T program being developed for patients with relapsed or refractory T-cell acute lymphoblastic leukemia and T-cell lymphoblastic lymphoma (T-ALL/LBL). The accumulation of U.S. FDA expedited development designations for WU-CART-007, together with active clinical studies evaluating safety and efficacy across patient populations, highlights sustained regulatory engagement and reinforces the program’s clinical momentum in a high-unmet-need setting. At the same time, Wugen’s current intellectual property footprint—still relatively limited and partly weakened by lapsed assets—appears poised for strengthening as pending filings mature and the platform evolves, making the company a noteworthy case study in the fast-moving allogeneic CAR-T landscape explored in our latest patent report.
Press contact
contact@knowmade.fr
Le Drakkar, 2405 route des Dolines, 06560 Valbonne Sophia Antipolis, France
www.knowmade.com
About the author
Fabienne Massa, PhD., works for KnowMade in the field of Biotechnology and Life Sciences. She holds a PhD in Molecular and Cellular Biology from the IPMC (Sophia Antipolis, France). She also holds a Master of Business Management from IAE (Nice, France) and she previously worked in the pharmaceutical industry.
About KnowMade
KnowMade is a technology intelligence and IP strategy consulting company specialized in analyzing patents and scientific publications. The company helps innovative companies, investors, and R&D organizations to understand competitive landscape, follow technological evolutions, reduce uncertainties, and identify opportunities and risks in terms of technology and intellectual property.
KnowMade’s analysts combine their strong technology expertise and in-depth knowledge of patents with powerful analytics tools and methodologies to turn patent information and scientific literature into actionable insights, providing high added value reports for decision makers working in R&D, innovation strategy, intellectual property, and marketing. Our experts provide prior art search, patent landscape analysis, freedom-to-operate analysis, IP due diligence, and monitoring services.
KnowMade has a solid expertise in Compound Semiconductors, Power Electronics, Batteries, RF Technologies & Wireless Communications, Solid-State Lighting & Display, Photonics, Memories, MEMS & Sensors, Semiconductor Packaging, Medical Devices, Medical Imaging, Microfluidics, Biotechnology, Pharmaceutics, and Agri-Food.
